Catch the gaps that trigger FDA deficiency letters
FDA reviewers flag the same preventable issues — incomplete sections, unsupported claims, non-compliant labeling. FDA Reviewer runs those same checks on your documents before you submit.
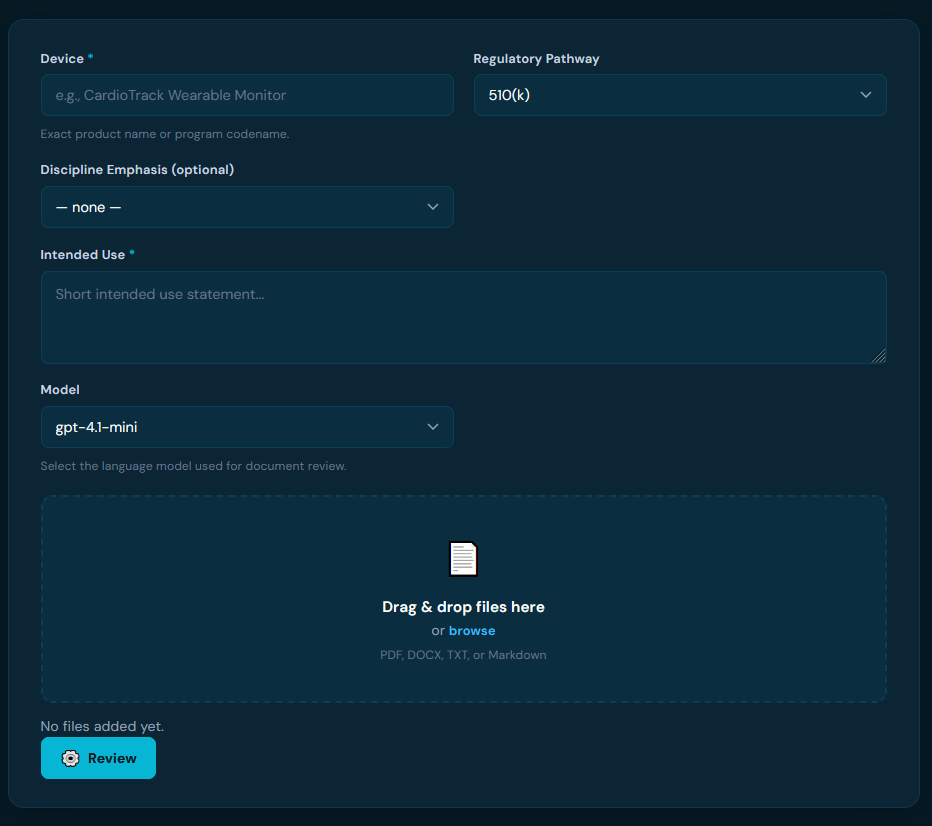
FDA reviewers flag the same preventable issues — incomplete sections, unsupported claims, non-compliant labeling. FDA Reviewer runs those same checks on your documents before you submit.

Not a generic AI assistant. A purpose-built review engine calibrated to FDA premarket pathways, guidance documents, and submission structure.
Reads your documents the way an FDA reviewer would — section by section, mapped against the guidance applicable to your specific pathway and device class.
Surfaces the most common causes of FDA additional information requests before you submit — missing data, incomplete comparisons, unsupported performance claims.
Pinpoints the exact sections, paragraphs, and tables that fall short of FDA expectations — with specific language recommendations, not just flags.
A single compliance score per document, broken down by section — so your team knows exactly where to focus revision effort before the next review cycle.
Set your device classification, submission type, and intended use once. Every analysis is calibrated to your regulatory pathway — 510(k), PMA, or De Novo.
Download a structured review package formatted for internal QA sign-off, team review, or regulatory consultant handoff — ready in minutes.
A single deficiency response can add 3–6 months to your review clock. FDA Reviewer surfaces the same gaps an FDA reviewer would flag — before your submission leaves your desk.
Start your first reviewDrag and drop your FDA submission files — PDFs, Word docs, spreadsheets. We handle all formats.
Tell us your device class, intended use, and target market. The AI tailors its analysis to your specific submission type.
Our models cross-reference your documents against current FDA guidance, predicate devices, and regulatory databases.
Get a structured review report with findings, risk ratings, suggested edits, and a compliance summary in minutes.
Regulatory teams that catch gaps internally move faster through FDA review — fewer deficiency letters, shorter response cycles, and more predictable timelines.
Tell us about your submission workflow and we'll scope a pilot around your documents and device type.
From first 510(k) to enterprise-scale submission portfolios — we scope every pilot around your actual regulatory workflow.
We'll get back to you within 24 hours.
Thanks for reaching out. Our team will be in touch within 1 business day with next steps and a personalized demo.